|
4/6/2024 0 Comments Periodic table molar mass cl2Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. Element Polonium (Po), Group 16, Atomic Number 84, p-block, Mass 209. Thus, the molar mass of bilirubin can be expressed as 584.05 g/mol, which is read as “five hundred eighty four point zero five grams per mole. Created by video journalist Brady Haran working with chemists at The University of Nottingham. 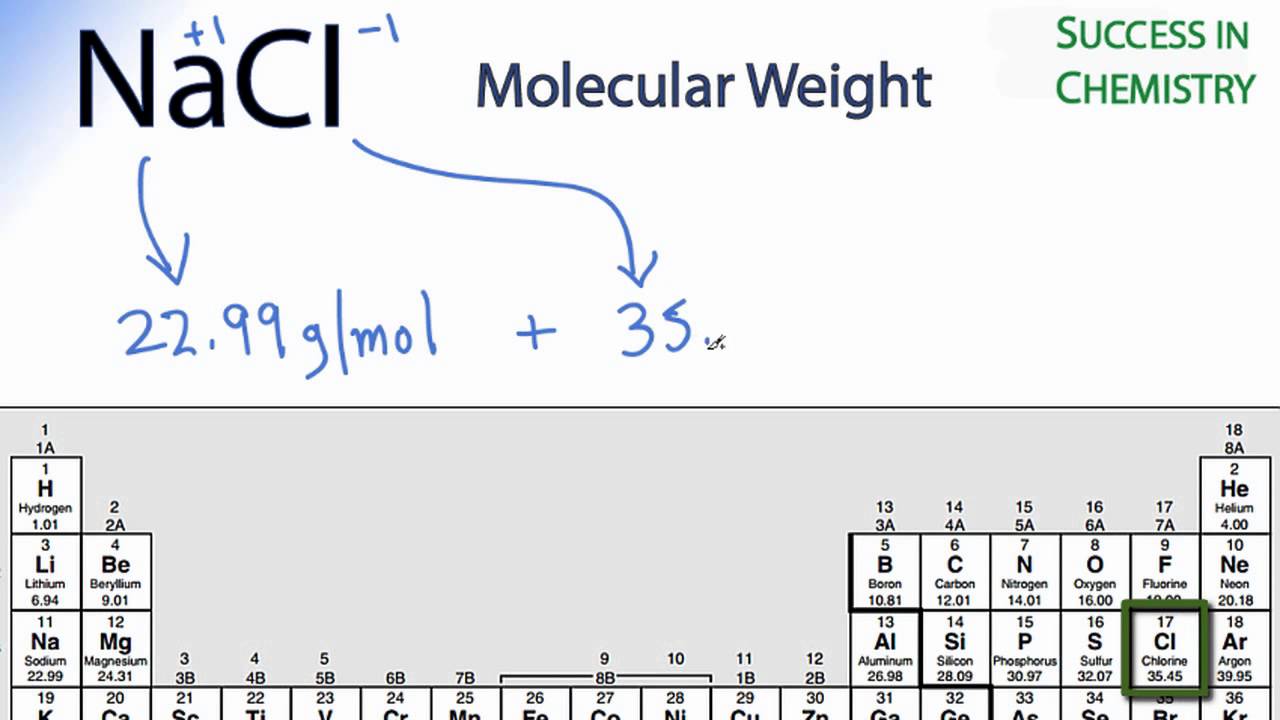
H2 + Cl2 HCl H2 + I2 HI H2 + Br2 HBr H2 + F2 HF Calculators. using the periodic table, find the atomic mass in g/mol of each element (the molar mass of an element is equal to its atomic mass). The division sign (/) implies “per,” and “1” is implied in the denominator. The molar mass and molecular weight of H2 (Dihydrogen) is 2.016. For example, the molar mass of Ba(OH) 2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: 1 Ba molar mass:īecause molar mass is defined as the mass for 1 mol of a substance, we can refer to molar mass as grams per mole (g/mol). In formulas with polyatomic ions in parentheses, the subscript outside the parentheses is applied to every atom inside the parentheses. The mass of 1 mol of bilirubin is 584.69 g.īe careful when counting atoms. Multiplying the molar mass of each atom by the number of atoms of that type in bilirubin’s formula and adding the results, we get.This is repeated until there is a term for each isotope. The weighted average is determined by multiplying the percent of natural abundance by the actual mass of the isotope. 
Summing the molar masses of the atoms in the NaCl formula unit gives The atomic mass of an element is the weighted average of the atomic masses of the naturally occurring isotopes of that element. 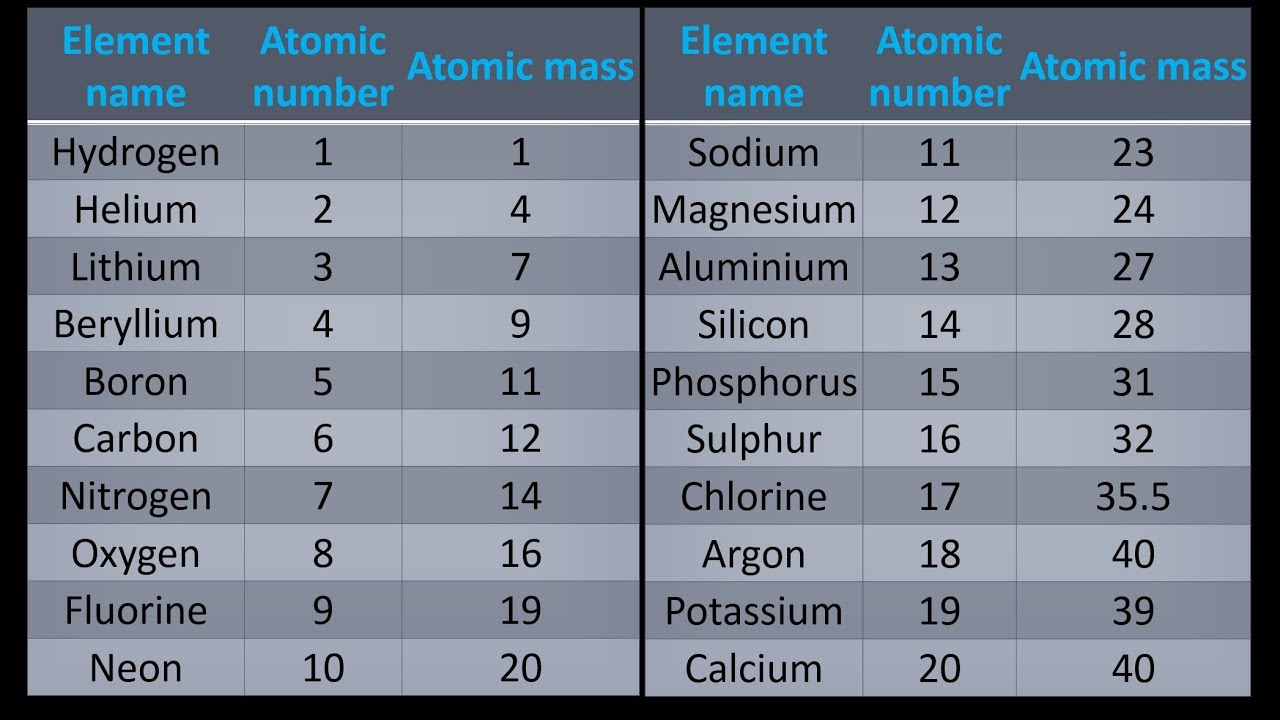
To find the mole fraction and percentage of each element in CoCl2, divide each total from step 3 by the total molar mass found in step 4: Mole Fraction. bilirubin (C 33H 36N 4O 6), the principal pigment present in bile (a liver secretion) Finally, add together the total mass of each element to get the molar mass of CoCl2: 58.933195 g/mol + 70.906 g/mol 129.839195 g/mol.What is the mass of 1 mol of each substance? \): Moles to Mass Conversion with Compounds
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
